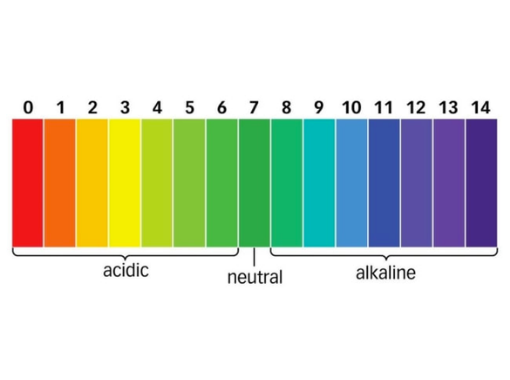
pH is a measure of how acidic a solution is. For dilute solutions the pH scale ranges from pH 0 to pH 14, pH seven is in the middle and is therefore the neutral point; pH’s less than 7 are acidic and pH’s greater than 7 are alkaline.
The pH scale is a measure of the concentration of hydrogen ions (measured as moles/litre) present in the water. The lower the pH reading the greater the concentration of hydrogen ions.
If you know the pH of the water it is easy to determine the concentration of hydrogen ions that are present.
– At pH 7 (Neutral pH) the concentration of hydrogen ions present in the solution is 1 x 10-7 moles per litre.
– At pH 2 (Highly acidic) a much greater concentration of hydrogen ions are present. At this pH the concentration of hydrogen ions present in the solution is 1 x 10-2 moles per litre.
– At pH 12 (Highly alkaline) a much lower concentration of hydrogen ions will be present. At this pH the concentration of hydrogen ions present in the solution is 1 x 10-12 moles per litre.
pH = -log (hydrogen ions concentration expressed as moles/litre).
Note:
Alkalinity is not the opposite of Acidity. Highly alkaline is the opposite of highly acidic.
Alkalinity is a measure of a solutions ability to resist a change to its pH. Commonly alkalinity is provided by the presence of Calcium and Magnesium Carbonates. Waters with high levels of alkalinity are termed hard (have ability to form scale and difficult to make a lather with soap) and waters with low alkalinity are termed soft (easy to form lather).
What does this mean?
The pH scale is not a linear scale; it is a log scale. Small changes in pH correspond to relatively large concentration changes.
The majority of the measuring scales that we encounter are linear scales and we have become attuned to interpreting linear scales, making it difficult for us to appreciate the magnitude of difference represented by small changes in pH (a log scale).
So to put the difference between a log scale and a linear scale into context, lets consider a car travelling along a motorway.
The speed limit on the motorway is 70 miles per hour, if the car travelled at 77 miles per hour it would be 10 % over the legal limit. Some people might consider this to be an acceptable level of compliance with the law.
Now let’s consider water with a pH of 9 and a second water with a pH of 9.9 (only 10% difference in pH units)
At pH 9 the hydrogen ion concentration is 1 x 10-9 moles/litre. At pH 9.9 the hydrogen ion concentration 1 x 10-9.9 moles per litre, the difference in hydrogen ion concentration is 1 x 10-0.9 moles per litre.
This means that at pH 9.9 the concentration of hydrogen ions is 87 % lower than at pH 9. Few if any would find this level of compliance as acceptable.
Small changes in pH correspond to large changes in water chemistry and can have a devastating effect on the health of the aquatic environment.
Natural Range
The majority of our inland lakes, canals, streams and ponds have a pH in the range 6 to 9. Surface water bodies with naturally occurring pH values outside of this range are less common, but they do exists, e.g. In upland moorland peaty areas surface water pH values may be in the range 5 to 6 and is some types of peat bog may fall in the range pH 3 to 4.
Man Made pollution has the ability to lower the pH of a water body to a value that is outside of a typical natural range.
For example:
Acidic
– Acid rain formed by the mixing of sulphur and nitrous gasses with rainwater, can cause the pH of rainwater to become highly acidic (pH 2 to 3).
– Metal Rich acidic waters discharged from mines. In addition to detrimental effects of low pH, these waters can also contain a variety of toxic metals. Increasing the hazard associated with their formation.
Alkaline
– A variety of industrial trade effluents that have highly alkaline pH, frequently associated with the use of either lime or caustic soda.
– Concrete wash water formed by the interaction of fresh concrete with water or the interaction of water with reactivated concrete. Reactivation is caused by either cutting or crushing of the concrete causing a fresh surface of concrete to be exposed and become wetted, causing the water to become highly alkaline. Note: Calcium Oxide (free lime in concrete) plus water forms Calcium Hydroxide (highly alkaline).
What are the typical pH limits that are applied to discharge into surface water?
Typically permitted discharges into surface water courses are restricted to a pH range of between 6 and 9. The discharge of water outside of these limits are almost certainly to be detrimental to the environment and will not be tolerated by the Regulator.
A discharge permit will include the general statement that the discharge must not cause harm/pollution to the environment. When assessing the potential for the discharge to cause harm there is a need to take the pH and volume of the receiving water course relative to the volume of the discharge into consideration.
Where ever possible the pH of the discharge water should match the pH of the receiving water. Limiting the potential for mixing of the two waters to cause the pH of the water course to change.
If the receiving water course is of low volume and a water of different pH is discharged then it is possible that the discharge of even small volumes of water with differing pH can be a cause of concern.
For example:
– A water course with a flow rate of 10 m3/hr and a pH of 6.0.
– A man made discharge of 60 m3/hr (3″ Pump) with a pH of 9.0, upper range of values stipulated within a typical environmental permit.
The discharge will change the pH of the receiving water course, the pH of the combined flow will have a pH of 6.8. A change of 0.8 pH Units.
To put the significance of this pH change into context, let’s consider the pH of our blood. Our body regulates the pH of our blood within narrow limits. The pH of our blood is maintained within a pH of 7.35 to 7.45, i.e. a variation of only 0.1 pH units. If the pH of our blood varied by 0.8 pH units irreversible cell damage would occur, poor health and possibly death would result.
If the manmade discharge is of an intermittent nature; for example, intermittent pumped discharge, there is a potential for rapid fluctuations in the pH of the water course to occur and whilst the pH of the water course remains within a typical natural range the rate of change could be excessive causing sensitive species to become stressed with fatalities among weaker members of the population occurring.
What effects can abnormal pH conditions have on the ecology?
Most freshwater lakes, streams, ponds canals etc have a pH in the range 6 to 8. A deviation in pH outside of this range will cause a decrease in both the number of individual species and the health of the surviving populations. Very few organisms can exist in waters with a pH of less than 3 or greater than 11.
Low pH
At pH 5.0, non-desirable species of plankton and mosses may dominate, fish such as small mouth bass may disappear.
Below pH 4.5, water may become devoid of fish. Toxic metals such as Aluminium may become soluble, causing fish to produce excessive concentrations of mucus and suffocate, fish may suffer chronic stress, loose body weight, becoming unable to compete for food and/or habitat. Eggs may have reduced fertility and larvae may develop abnormally.
Below pH 3.0, with the exception of a limited number of specialist organisms the water will be devoid of life.
High pH
At pH 9.6, the gills, eyes and skin of fish may be attacked reducing their protective abilities causing stress and fatalities, fish may be unable to dispose of their metabolic wastes.
Above pH 11, the water will be devoid of life.
Summary
An aquatic ecosystem such as a surface water body operates healthily over a narrow pH range. Man’s activities have the potential to:
1. Change the pH to such a degree that it falls outside of its natural range
2. Cause rapid fluctuations in pH.
Either of the above events can cause wildlife to become stressed and may cause fatalities. Reducing both population numbers and the health of any surviving members.
The original article can be found at Watery News